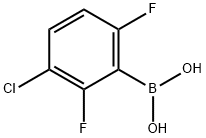
3-Chloro-2,6-difluorophenylboronic acid
3-Chloro-2,6-difluorophenylboronic acid
CAS: 1031226-45-5
Molecular Formula: C6H4BClF2O2
3-Chloro-2,6-difluorophenylboronic acid - Names and Identifiers
| Name | 3-Chloro-2,6-difluorophenylboronic acid |
| Synonyms | 3-CHLORO-2,6-DIFLUOROPHENYLBORONIC ACID 3-Chloro-2,6-difluorophenylboronic acid |
| CAS | 1031226-45-5 |
3-Chloro-2,6-difluorophenylboronic acid - Physico-chemical Properties
| Molecular Formula | C6H4BClF2O2 |
| Molar Mass | 192.3555664 |
| Storage Condition | 2-8℃ |
| Sensitive | Irritant, Refrigerate |
| MDL | MFCD04038753 |
3-Chloro-2,6-difluorophenylboronic acid - Introduction
3-Chloro-2, 6-difluorophenylboronic acid is an organic compound with the formula C6H3ClF2BO2. A description of the properties, uses, preparation and safety information of the compound is given below:
Nature:
3-Chloro-2, 6-difluorophenylboronic acid is a colorless to pale yellow crystal or powder with weak acidity. It is soluble in organic solvents such as ethanol, dimethylformamide and the like at room temperature.
Use:
3-Chloro-2, 6-difluorobenzeneboronic acid is one of the reagents commonly used in organic synthesis. It can be used to catalyze the formation of carbon-silicon bonds and participate in a variety of reactions, such as the formation of borohydride reagents, the esterification of phenylboronic acid, and the formation of C- C bonds in organic synthesis. In addition, it can also be used as an acid catalyst for aromatic hydrocarbons.
Preparation Method:
The preparation method of 3-chloro-2, 6-difluorobenzeneboronic acid is usually obtained by introducing a fluorine atom at the ortho position of a halogen acid (such as ferric chloride), and then reacting with boric acid. Specific methods of preparation can be found in the manual of organic chemical synthesis.
Safety Information:
Since 3-chloro-2, 6-difluorophenylboronic acid is an organic compound, it is necessary to pay attention to safety during use and operation. It can be irritating to the eyes, skin, and respiratory tract, so contact should be avoided. When using it, you should wear appropriate personal protective equipment, such as laboratory gloves, laboratory glasses, protective clothing, etc. In addition, the inhalation of its dust should be avoided to avoid damage to the respiratory system. When stored, it should be kept away from fire and oxidizing agents and stored in a dry place. In order to ensure safety, it is best to use it in an experienced chemical laboratory and follow the relevant operating procedures.
Nature:
3-Chloro-2, 6-difluorophenylboronic acid is a colorless to pale yellow crystal or powder with weak acidity. It is soluble in organic solvents such as ethanol, dimethylformamide and the like at room temperature.
Use:
3-Chloro-2, 6-difluorobenzeneboronic acid is one of the reagents commonly used in organic synthesis. It can be used to catalyze the formation of carbon-silicon bonds and participate in a variety of reactions, such as the formation of borohydride reagents, the esterification of phenylboronic acid, and the formation of C- C bonds in organic synthesis. In addition, it can also be used as an acid catalyst for aromatic hydrocarbons.
Preparation Method:
The preparation method of 3-chloro-2, 6-difluorobenzeneboronic acid is usually obtained by introducing a fluorine atom at the ortho position of a halogen acid (such as ferric chloride), and then reacting with boric acid. Specific methods of preparation can be found in the manual of organic chemical synthesis.
Safety Information:
Since 3-chloro-2, 6-difluorophenylboronic acid is an organic compound, it is necessary to pay attention to safety during use and operation. It can be irritating to the eyes, skin, and respiratory tract, so contact should be avoided. When using it, you should wear appropriate personal protective equipment, such as laboratory gloves, laboratory glasses, protective clothing, etc. In addition, the inhalation of its dust should be avoided to avoid damage to the respiratory system. When stored, it should be kept away from fire and oxidizing agents and stored in a dry place. In order to ensure safety, it is best to use it in an experienced chemical laboratory and follow the relevant operating procedures.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 3-CHLORO-2,6-DIFLUOROPHENYLBORONIC ACID Visit Supplier Webpage Request for quotationCAS: 1031226-45-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-CHLORO-2,6-DIFLUOROPHENYLBORONIC ACID Request for quotation
CAS: 1031226-45-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1031226-45-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: 3-CHLORO-2,6-DIFLUOROPHENYLBORONIC ACID Visit Supplier Webpage Request for quotationCAS: 1031226-45-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-CHLORO-2,6-DIFLUOROPHENYLBORONIC ACID Request for quotation
CAS: 1031226-45-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1031226-45-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History

